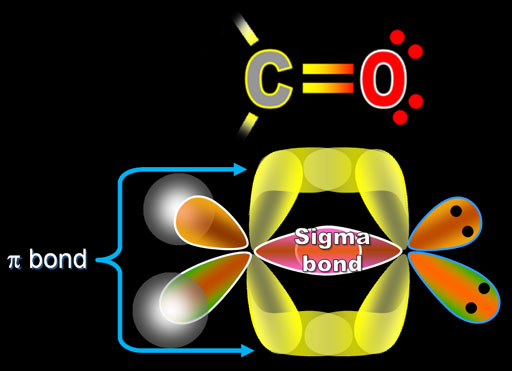
The pi bond between the two carbon atoms has one node in the plane of the molecule. The sigma bond between the two carbon atoms does not have a node in the plane of the molecule. The wave with a single node has higher energy. If your workstation is enabled for JCE Software, you will see two videos below which compare the behavior of a standing wave with zero nodes versus a standing wave with one node (otherwise, see the drum animation below). The pi bond can be thought of as a standing wave with a single node in the plane of the molecule. Each of the two electrons in the pi bond (π bond) exists both above and below the plane of the four H atoms and the two C atoms. The pi bond (π bond) has two halves-one above the plane of the molecule, and the other below it. This is called a pi bond, Greek letter π. A second carbon-carbon bond is formed by the overlap of these two remaining p orbitals. The sp2 hybrid orbitals on each carbon atom involve the 2 s and two of the 2 p orbitals, leaving a single 2 p orbital on each carbon atom. By selecting N8 HOMO, you can see the pi orbital represented by the two lobes. This is actually sigma bonding between C-C and some sigma-like bonding around the Hs as well. To view the sigma bonding orbital, select N6. These overlap sideways to form a π bond, also shown in gray. Two p orbitals, one on each C atom, are shown in gray. 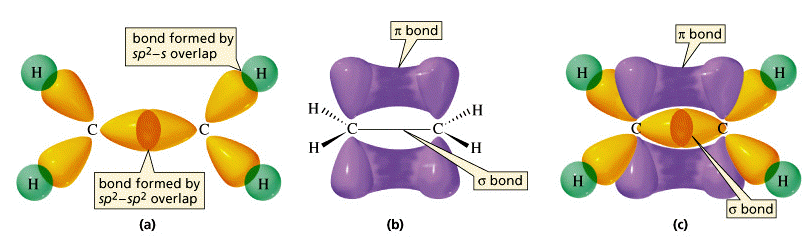
Two of these overlap directly between the carbon atoms to form the σ bond. Three sp 2 hybrids around each carbon atom are indicated in color.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
